Main Second Level Navigation
Mar 1, 2022
RNA Map of the Mammalian Cell Nucleus Reveals New Insights into Gene Regulation
Research
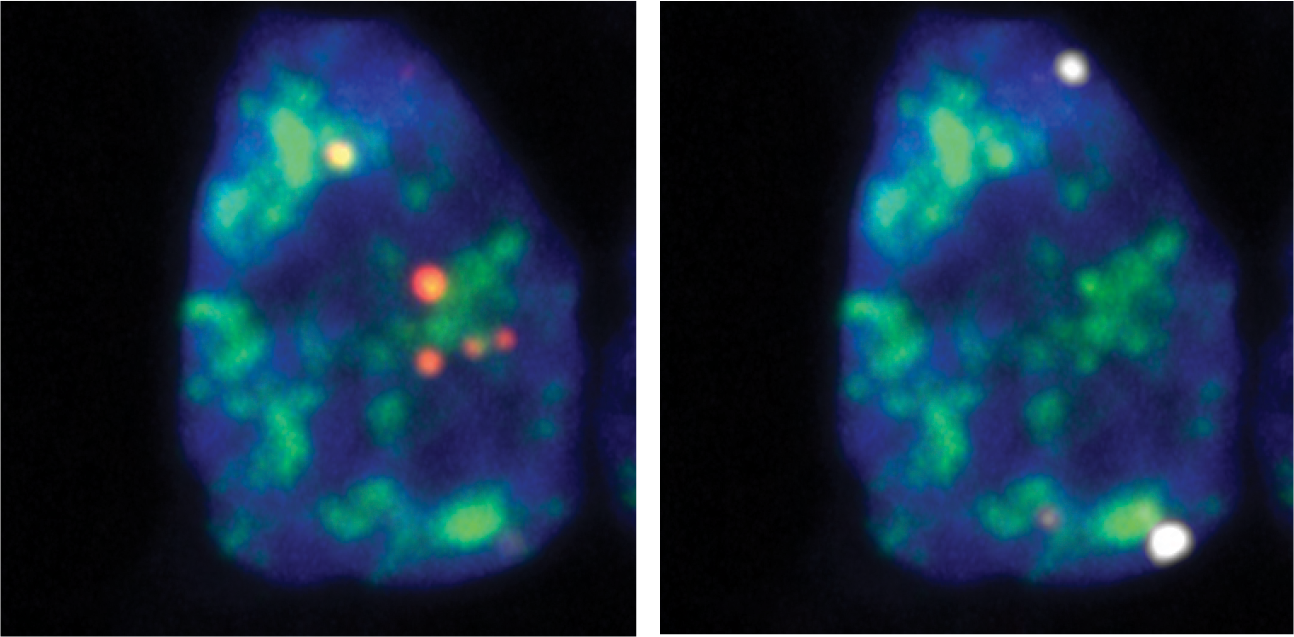
Barutcu, Wu et al. Mol Cell
Microscopy images of cell nuclei with fluorescently labeled RNA transcripts which contain retained introns. On the left are shown transcripts associated with nuclear speckles, while lamina-associated transcripts are shown on the right.



