Main Second Level Navigation
Oct 25, 2021
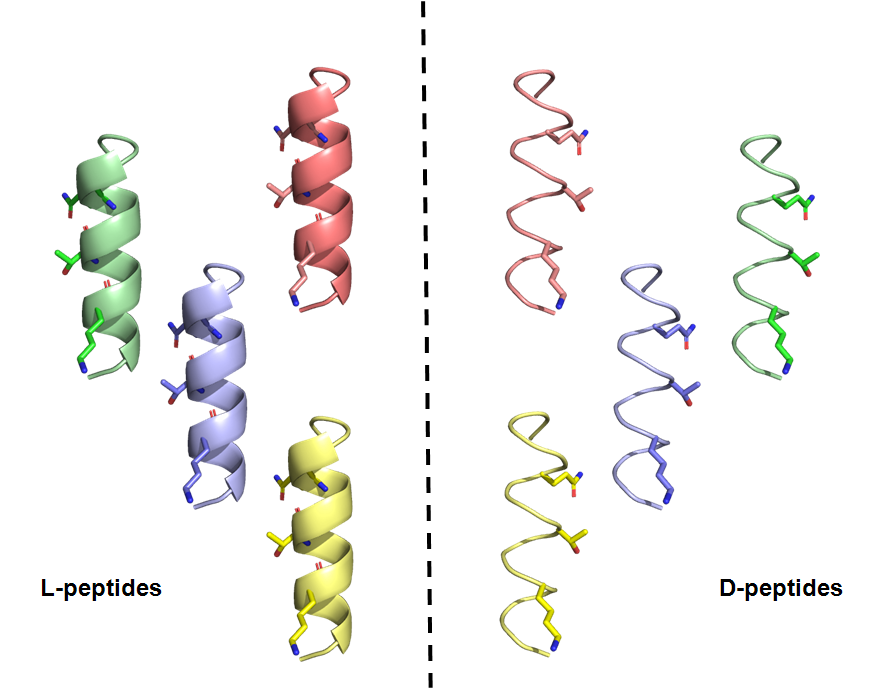
Donnelly Centre investigators create mirror-image peptides capable of neutralizing Sars-CoV-2
Protein Engineering, Research
Dr. Pedro Valiente
Mirror-image peptides (D-peptides on the right) engineered by Donnelly Centre investigators neutralised Sars-CoV-2 and prevented infection of cultured human cells.




